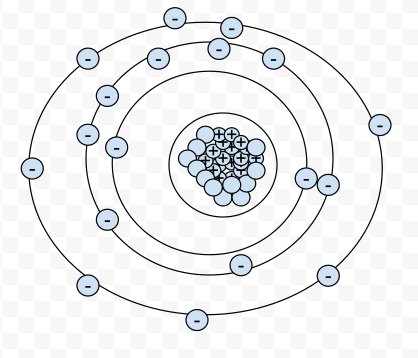
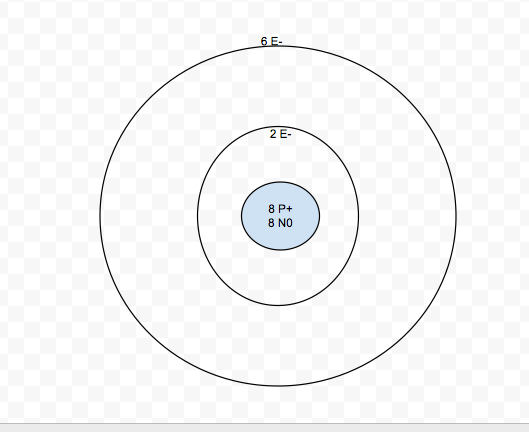
3-21-13
Anything that has mass and takes up space is defined as matter. Matter is made up of neutrons, protons, electrons, quarks, gluons and more. Then you cannot see everything in the matter but most things you can see.
3-22-13
Atomic Number= 4
Atomic Mass= 9
Element= Beryllium
Protons= 4
Neutrons= 5
Electrons= 4
Atomic Mass= 9
Element= Beryllium
Protons= 4
Neutrons= 5
Electrons= 4
3-25-13
10 is atomic number. 20 is atomic mass. The name is Neon.
3-26-13
The warm-up. Argon! Noble Gas! 18 Protons and electrons. Level three energy. 22 Protons.
3-27-13
Neon
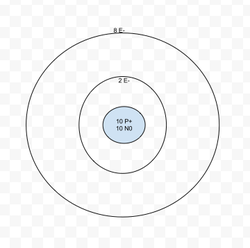
10
Atomic Mass: 20.17
Atomic Number: 10
Neon
Atomic Mass: 20.17
Atomic Number: 10
Neon
3-28-13
This element is Sodium. No the element is not stable. This would form a positive ion.
4-1-13
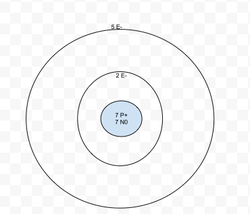
Nitrogen is not stable. It will become a negative Ion. Nitrogen has 5 valence electrons.
4-4-13
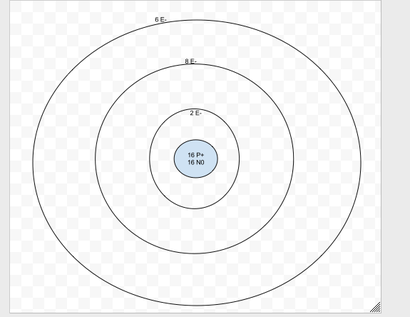
It has six valence electrons. It will form a negative Ion. it will bond with beryllium.
4-5-13
Magnesium. Carbon has 4 valence electrons. Carbon is not stable. It will bond with Silicon.
4-8-13
The element is Nitrogen. It is not stable. It will bond with Boron.
4-15-13
The answer is C, the continuous motion of air and water distribute the sun's energy.
4-29-13
No, it is not stable. It has 6 valence electrons and 8 protons and nuetrons. It forms a negative ion.
4-30-13
There are 7 atoms in the reactants and 6 on the products.
There are 3 elements on both sides.
The equation does not support the conservation of mass.
There are 3 elements on both sides.
The equation does not support the conservation of mass.
5-1-13
_Y + _R2 = _Y2R3
4Y + 3R2 = 2Y2R3
4Y + 3R2 = 2Y2R3