3-25-13
Valence Electrons
Valence electrons are how many electrons are on outer level. Noble gases have no charge so they are stable. Seven energy levels.
Notes
Electrons are ordered in layers. Layers of the electron cloud are called energy levels or shells. There are a maximum of seven energy levels.
The first energy level can have two electrons. The rest can have eight. Number of electrons in the outer energy layer controls how or if atoms react with each other.
Valence Shell is the highest energy level that an atom has.
Valence Electrons are how many electrons are how many electrons you have on your outermost shell.
The first energy level can have two electrons. The rest can have eight. Number of electrons in the outer energy layer controls how or if atoms react with each other.
Valence Shell is the highest energy level that an atom has.
Valence Electrons are how many electrons are how many electrons you have on your outermost shell.
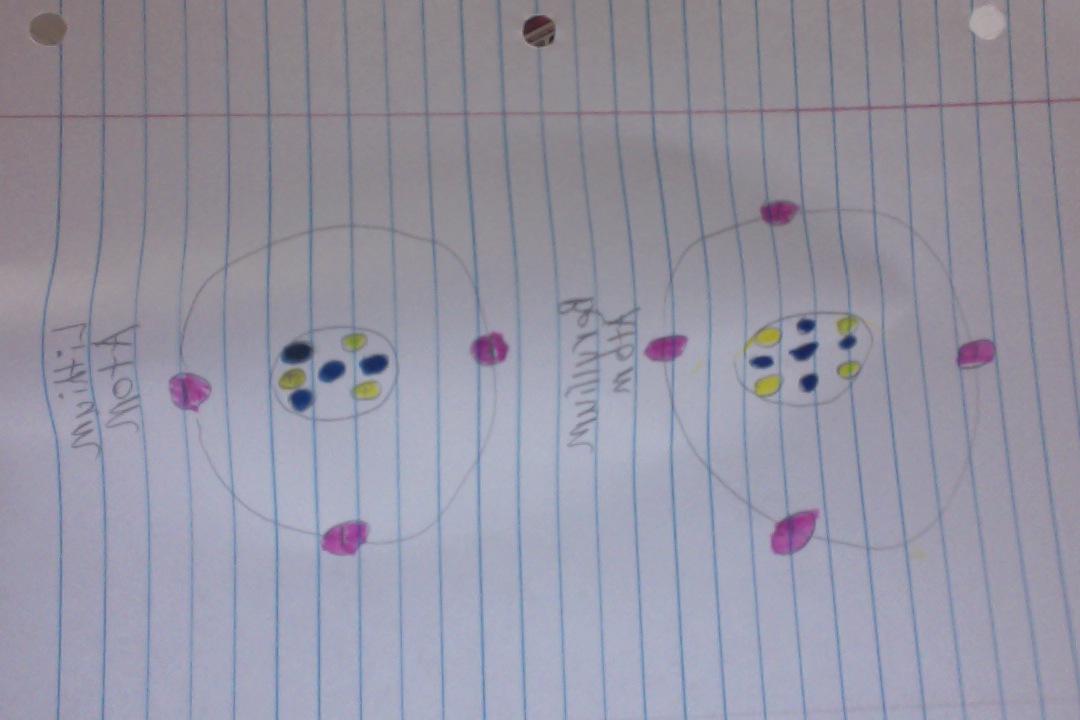
Boron
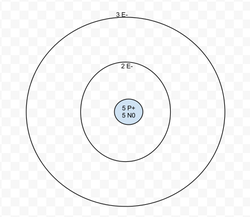
P+=5
N0=6
E-=5
Valence Electrons= 3
Energy level isn't full.
N0=6
E-=5
Valence Electrons= 3
Energy level isn't full.
Nitrogen
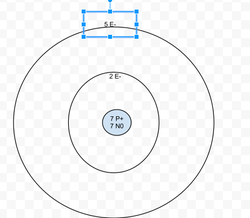
Protons: 7
Neutrons: 7
Electrons: 7
Valence Electrons = 5
Last energy level isn't full
Neutrons: 7
Electrons: 7
Valence Electrons = 5
Last energy level isn't full
Aluminum
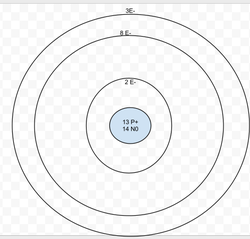
Protons: 13
Neutrons: 14
Electrons: 13
Valence Electrons = 3
Last energy level isn't full
Neutrons: 14
Electrons: 13
Valence Electrons = 3
Last energy level isn't full
Phosphorous
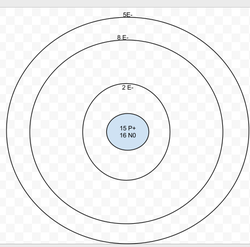
Protons: 15
Neutrons: 16
Electrons: 15
Valence Electrons = 5
Last energy level isn't full
Neutrons: 16
Electrons: 15
Valence Electrons = 5
Last energy level isn't full
Calcium
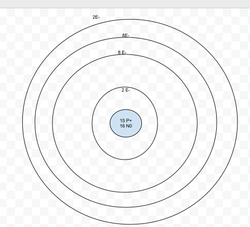
Protons: 20
Neutrons: 20
Electrons: 20
Valence Electrons = 2
Last energy level isn't full
Neutrons: 20
Electrons: 20
Valence Electrons = 2
Last energy level isn't full
Reactivity
Atoms tend to combine so that their valence shells are full.
If a neutral atoms valence shell is full then it will not react with other atoms. If there is space available then the atom will react with other atoms. Some atoms lose electrons and become positive ions. Some atoms gain electrons to become negative ions.
If a neutral atoms valence shell is full then it will not react with other atoms. If there is space available then the atom will react with other atoms. Some atoms lose electrons and become positive ions. Some atoms gain electrons to become negative ions.