3-21-13
Chemistry
Element- Substance that cannot be broken down by chemical means. Has specific amounts of protons.
Atom- Smallest possible particle of an element. Makes up everything around you.
Atom- Smallest possible particle of an element. Makes up everything around you.
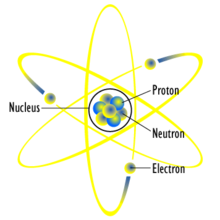
Proton determines elements name
Neutron determines atomic mass
Electrons cause chemical reactions
Electron Cloud is where atoms are found
Neutron determines atomic mass
Electrons cause chemical reactions
Electron Cloud is where atoms are found
Atomic Number
Protons
Electrons
Mass number minus the:
Atomic number equals the:
Neutrons
Protons
Electrons
Mass number minus the:
Atomic number equals the:
Neutrons
If the atom nucleus is one inch then the electron cloud is 1.6 miles
One Neutron equals One proton equals One AMU which equals 1836 Electrons
One Neutron equals One proton equals One AMU which equals 1836 Electrons
Questions
- In the nucleus is where the mass is found.
- The electron cloud has the largest volume.
- The nucleus is positive.
- The electron cloud is negatively charged.
- The overall charge of the nucleus is positive and the electron cloud is negative which cancel each other out and keeps the atom stable and together.
- The electron to proton ratio is the same in both (1:1). They both have all the subatomic particles.
- Hydrogen has one of everything. Hydrogen has two of everything.
Atoms
- Hydrogen
- Helium
- Lithium
- Beryllium
Proton #
|
Neutron #
|
AMU #
|
Electron #
|
Charge
|
Questions pt 2.
NONE
NONE
NONE
NONE
NONE
NONE
NONE
NONE
NONE