3-28-13
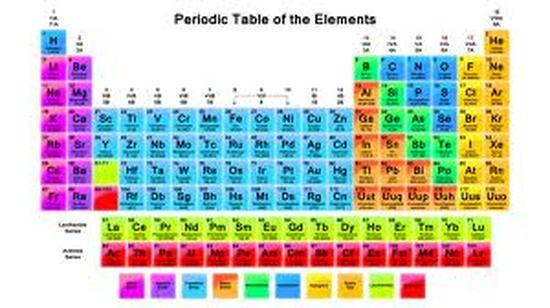
Periodic Table
Chart of all the elements. Based on physical and chemical properties. Electrons orbit the nucleus in electrons cloud. Level 1 - 2 electrons. Level 2 - 8 electrons. Level 3 - 8 electrons. Valence electrons are the number of electrons in the last energy level.
Bohr Model
Bohr atomic models show energy levels and valence electrons.
Organization
7 Rows- Called Periods. Each period represents the number of occupied energy levels in each element in the row.
18 Columns- Called Families. Represents valence electrons. Also atoms in families share chemical and physical properties.
Staircase- Everything left of it is a metal with a few exceptions.
Metalloids- Boron, Silicon, Germanium, Arsenic, Antimony, Tellurium, Astatine.
18 Columns- Called Families. Represents valence electrons. Also atoms in families share chemical and physical properties.
Staircase- Everything left of it is a metal with a few exceptions.
Metalloids- Boron, Silicon, Germanium, Arsenic, Antimony, Tellurium, Astatine.
Properties of Metal
Metals are good at conducting heat and electricity. Metals are shiny. Metals are ductile( can be stretched). Metals are malleable (can be pounded into thin sheets). Chemical property includes reactivity with water.
Properties of Non-Metal
Non-metals poor conductors of heat and electricity. Non-metals are not ductile or malleable. Solid non-metals are brittle and easy to break. They are dull. Many non-metals are gases.
Properties of Metalloids
Metalloids have properties of both metal and non-metals. They are solids and can be shiny or dull. They conduct heat and electricity better than non-metals but not as well as metals. They are ductile and malleable.
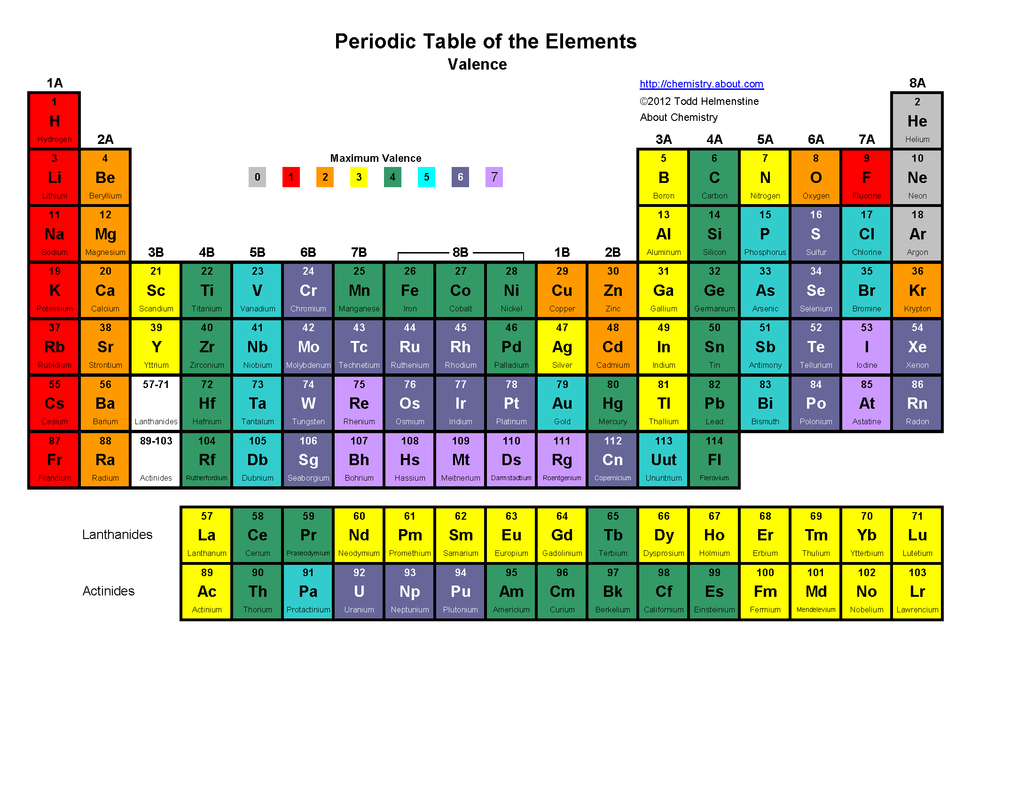
Groups
Group 1 - Alkali Metals. Will lose only electron in outer shell to become stable. Most reactive metal.
Group 2 - Alkali Earth Metals. Will lose two electrons in outer shell to become stable.
Group 17 - Halogens. Will gain one electron to become stable.
Group 18 - Noble Gases. Most stable elements. Outer shell full. All found in earths atmosphere.
Group 2 - Alkali Earth Metals. Will lose two electrons in outer shell to become stable.
Group 17 - Halogens. Will gain one electron to become stable.
Group 18 - Noble Gases. Most stable elements. Outer shell full. All found in earths atmosphere.