4-6-13
Volcano Lab
Comparative
Problem
Does the ratio of Baking soda to vinegar with the same amount of material used affect how much carbon dioxide is produced?
Research
Chemical Reaction: A chemical reaction is a process by which two or more chemical react to form one or more new completely different substance(s). There are several indicators of a chemical reaction. Bubbles is the most prominent one and that indicates that a gas is forming. other common ones is a change in temperature, color and acidity levels. All chemical reactions have to follow the law of the conservation of mass which means that there are the same amount of atoms on one side as there are on the other side and that only elements that appeared on the reaction side appear on the product side and there are no extras.
Volcanoes: Volcanoes are a natural geographical feature that forms at a subduction zone in a continental v ocean plate boundary or a oceanic v oceanic v oceanic plate boundary, though these often form islands and island arcs that are volcanoes. Volcanoes build up a substance called magma that comes from the mantle and when the pressure is great enough releases it all at one once in a gigantic eruption. Magma is lava but before it reaches the surface.
Baking Soda and Vinegar: Baking Soda and Vinegar are a pair of items that when combined form a chemical reaction. The resulting products include CO2 which is also known as carbon dioxide and is what will be used to measure my reaction. One of the other products is water which can also be measured for the resulting chemical reaction. The chemical formula is as follows: CH3COOH + NaHCO3 ---> CH3COONa + H2O + CO2
Volcanoes: Volcanoes are a natural geographical feature that forms at a subduction zone in a continental v ocean plate boundary or a oceanic v oceanic v oceanic plate boundary, though these often form islands and island arcs that are volcanoes. Volcanoes build up a substance called magma that comes from the mantle and when the pressure is great enough releases it all at one once in a gigantic eruption. Magma is lava but before it reaches the surface.
Baking Soda and Vinegar: Baking Soda and Vinegar are a pair of items that when combined form a chemical reaction. The resulting products include CO2 which is also known as carbon dioxide and is what will be used to measure my reaction. One of the other products is water which can also be measured for the resulting chemical reaction. The chemical formula is as follows: CH3COOH + NaHCO3 ---> CH3COONa + H2O + CO2
Citations
- "Chemical Reaction." Fact Monster. N.p., n.d. Web. 5 Apr. 2013. <http://webpath.follettsoftware.com/resource/viewurl?encodedUrl=plo2j1Ikxcj5uQozTp-kxWk-2OggZEc4XKww1zYu0NQ&version=1&userGUID=1DB26884-89B5-4741-A396-0B10CFF447F9&gv=1&gc=-164344351&appsignature=Destiny&appversion=10.5.6.0+%28RC6%29>.
- "About Volcanoes." About Volcanoes. N.p., n.d. Web. 5 Apr. 2013. <http://webpath.follettsoftware.com/resource/viewurl?encodedUrl=nyksLrFVtJi-9UbW173cmZHUzhDSzFoOEEhJY2Izc7c&version=1&userGUID=1DB26884-89B5-4741-A396-0B10CFF447F9&gv=1&gc=-164344351&appsignature=Destiny&appversion=10.5.6.0+%28RC6%29>.
- "Rocketology: Baking Soda Vinegar = Lift Off!" Rocketology: Baking Soda Vinegar = Lift Off! N.p., n.d. Web. 10 Apr. 2013. <http://www.sciencebuddies.org/science-fair-projects/project_ideas/Chem_p086.shtml?fave=no>.
Hypothesis
I believe that a ratio of 1:1 will form the most CO2 gases.
Independent Variable: The ratio
Dependent Variable: The amount of CO2 produced.
Independent Variable: The ratio
Dependent Variable: The amount of CO2 produced.
Materials
- Baking Soda
- Vinegar
- Balloon
- Flexible ruler
- Marker
Method
- Open both the vinegar and baking soda containment units.
- With the marker make a mark around the balloon that goes in a straight line and meets the start.
- Pour 1 cups of vinegar into the bottle. You mayor may not need a funnel for this step.
- Pour 16 tablespoons of baking soda into the balloon. This step requires the use of a funnel. This is because 16 tablespoons is equivalent to one cup.
- Now put the lip of the balloon over the opening of the bottle. However do not pour the baking soda in yet.
- Next sit the balloon straight up and let all of the baking soda pour from the balloon.
- Now the reaction will take place and the balloon will fill up. Once the bubbles in the bottle stop forming use the tape measure to measure the mark that you made around the balloon.
- Record results.
- Repeat steps 3-8 with the same ratio 2 more times.
- Repeat steps 3-9 with different ratios.
Data/Analysis
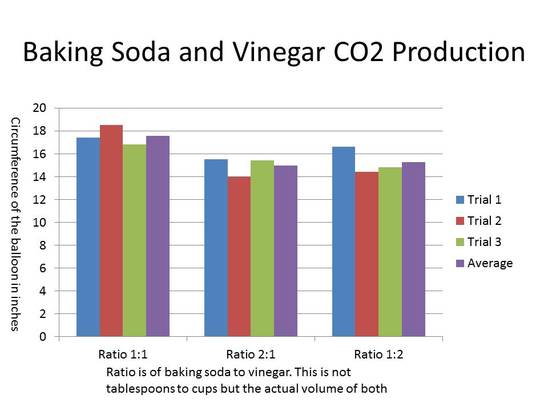
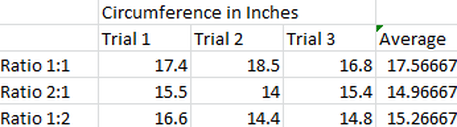
The data here shows that the ratio of 1:1 of baking soda and vinegar was the best. This is means that there is the same volume of both the baking soda and vinegar. The other two ratios were really close but were not close to the ratio of the 1:1 ratio.
Conclusion
My hypothesis was correct. When I mixed with the ratio of 1:1 it produced the most CO2. The data clearly shows that the 1:1 ratio produced more CO2 than the others. In fact,on average the 1:1 ratio produced an extra of 2 inches in circumference. The other ratios had a difference of less than a half-an-inch in circumference. The difference between these in separate trials is consistent as well and so has led me to believe that the ratio of 1:1 is the best. The reason for this being the most reactive has to do with the chemical equation. The chemical equation is CH3COOH + NaHCO3 ---> CH3COONa + H2O + CO2. This means that there should be one molecule of baking soda and one molecule of vinegar to react and form the new elements that we measure (i.e. CO2). If the total volume stays the same but there are different ratios then this means that the amount of CO2 will be equivalent to the material that has the least amount of mass.